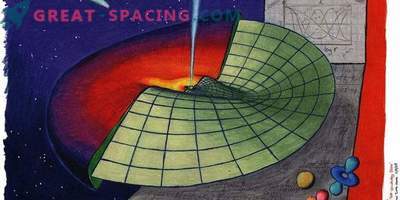
You may not understand anything in physics, but you should have heard about Schrödinger's thought experiment, where the cat is placed in a box with a radioactive element and it can be both alive and dead. This is a strange phenomenon created by quantum mechanics.
Recently, physicists from the University of Exeter (England) discovered that a similar similarity can be seen in temperatures: objects can have two temperatures at the quantum level. This strange quantum paradox is the first completely new relation of quantum uncertainty that will be formulated over decades.
Another Heisenberg Principle
In 1927, the German physicist Werner Heisenberg made a postulate: the more accurately you measure the position of a quantum particle, the less accurately you will understand its momentum and vice versa. This rule is now called the Heisenberg Uncertainty Principle.
The new quantum uncertainty, where the more accurately you know the temperature, the less you can say about energy and vice versa, has much greater implications for nanoscience studying incredibly tiny objects (smaller than nanometer). This principle will change how scientists measure the temperature of extremely small things, like quantum dots. In the 1930s. Heisenberg and Niels Bor have established a relationship between the uncertainty between energy and temperature on a non-quantified scale. The idea was that if you wanted to know the exact temperature of the object, it would be better to immerse it in a “tank” (a bath with water or a chamber with air) with a known temperature, allowing the body to slowly saturate with this temperature. This is called thermal equilibrium.
This thermal equilibrium is maintained by the object while the reservoir is constantly exchanging energy. As a result, the energy in the object moves up and down in infinitely small quantities, which makes impossible an exact determination. If you want to know the exact energy in the object, you will have to isolate it so that it cannot contact with anything. However, the insulation does not allow to accurately calculate the temperature using the tank. These restrictions make the temperature uncertain, and when moving to a quantum scale, the colors thicken even more.
New Uncertainty Ratio
Even if a typical thermometer has energy that rises and falls slightly, it is still detectable in a small range. But this does not work at the quantum level, where everything returns to the famous Schrödinger cat. This thought experiment suggested closing the cat in a box of poison, activated by the decay of a radioactive particle. According to the laws of quantum mechanics, a particle can decompose or not collapse at the same time. That is, until you open the box, the cat will be both alive and dead at the same time. This is a superposition phenomenon. The researchers used mathematics and theory to accurately predict how superposition affects the temperature calculation of quantum objects. It turns out that a quantum thermometer will be in the superposition of energy states at the same time, which leads to temperature uncertainty.
In our world, a thermometer can report that an object is between 31 and 32 degrees Fahrenheit. In the quantum case, the thermometer will say that the object is simultaneously endowed with both temperatures. Contacts between objects in a quantum scale are capable of creating superpositions and energy. The old uncertainty relation ignored these effects because they were unimportant for non-quantum objects. Now it is important if you need to determine the temperature index of a quantum dot.