Chemical engineers from Caltech have figured out why oxygen is being released from comets. It is also called O2 or molecular oxygen. This process was discovered in 2015 on comet 67P / Churyumov-Gerasimenko. It turned out that molecular oxygen levels were present in its atmosphere.
This gas is rather unstable in space, because it combines with hydrogen to become water, or with carbon and form carbon dioxide. O2 was found only in foggy areas with star formation. Researchers believed that molecular oxygen on a comet could freeze another 4.6 billion years ago and was released when the ice began to melt. But the question is still not removed, as it is believed that oxygen should react with other chemicals.
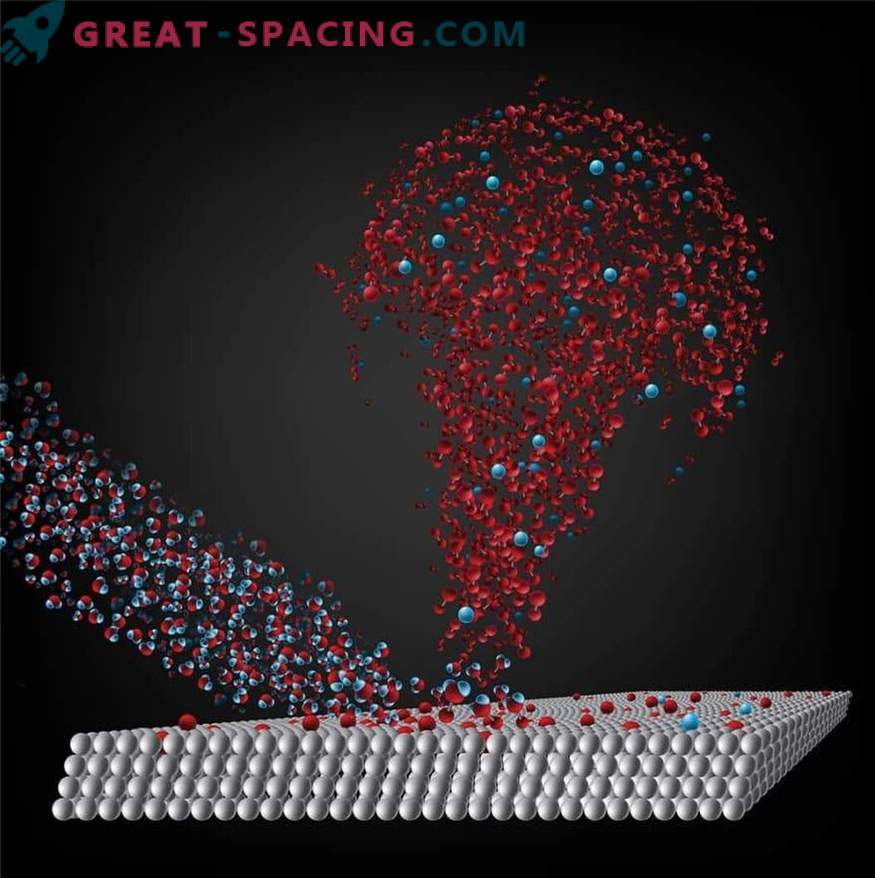
Laboratory experiments showed how to obtain molecular oxygen on the surface of comets
Scientists from the university began research with data from Rosetta, because the chemical reactions on the surface of the comet were similar to those that were carried out in laboratory conditions. Konstantinos P. Giapis was looking for a place in space where ions were accelerated from the surface. And he found them on the comet of Rosetta. In a new study with Yunhi Yao, they demonstrated that the comet could produce oxygen. Molecules of water vapor flowed out of the body as it approached the Sun and heated. Water molecules ionize, and the solar wind sends them back to the comet. When falling onto a surface that holds oxygen, the molecules “pick up” another oxygen atom and form O2.
That is, molecular oxygen on Rosetta may not have a connection with the primitive, but it can form in real time on the comet itself. A similar scheme can work on exoplanets, on which there is no life. This significantly influences how in the future researchers will search for objects suitable for study (search for extraterrestrial life).