The composition of the universe (the building blocks for each element of matter) is constantly changing and evolving due to the life and death of stars. Researchers report that interesting changes occurred in space, which greatly influenced the periodic table.
For 100 million years after the Big Bang, there was nothing but hydrogen, helium, and lithium. Then carbon, oxygen and other important components began to form. Now we live in the days of a full-fledged periodic table.
The periodic table helps people understand the elements of the Universe from the 1860s, when the Russian chemist Dmitry Mendeleev organized the chemical elements in a structured table. This order can be found in the chemical offices of any school, as well as in professional laboratories.

The number of elements at the age of the universe is 15 minutes
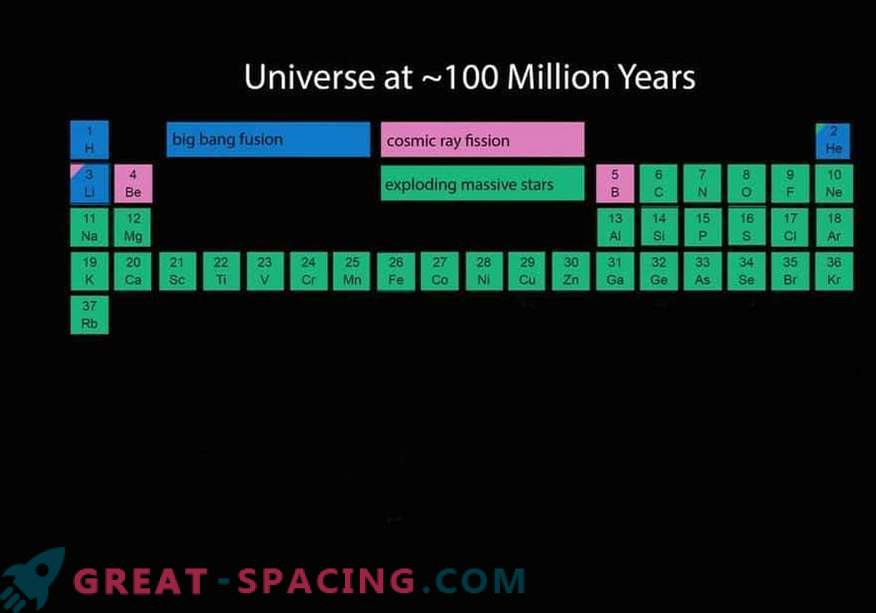
The number of elements at the age of the Universe is 100 million years.

The number of elements at the age of the Universe is 200 million years.

The number of elements at the age of the Universe is 8 billion years.
It is important to understand that most of the elements of the periodic table appeared in the stars. The table is constantly updated with new discoveries or in the case of synthetic elements created in laboratories around the world. Nucleosynthesis (the process of creating a new element) began with the Big Bang 13.7 billion years ago. The lightest elements (hydrogen and helium) were the first results of this event. But the heavier ones (almost everyone else in the table) are the result of the birth, evolution and death of stars.
Massive stars connect elements much faster than lower-mass stars. These giants merge hydrogen and helium into carbon, transforming it into magnesium, sodium and neon. Then such star objects die in a supernova explosion, releasing elements (from oxygen to silicon and selenium) into the surrounding space.
Stars with a smaller mass (like the Sun) combine hydrogen and helium in their own nuclei. Then helium merges with carbon, and after death the star becomes a white dwarf. These objects synthesize other elements when they explode or merge, where calcium and iron are pulled free. The fusion of neutron stars creates rhodium or xenon. So the death of existing celestial objects is constantly changing the composition of the elements in the universe.